As discussed here extensively, nothing in “evolution” makes any sense: “natural selection, fitness, speciation, human evolution, gradualism, divergence of character, UCD, TOL, etc. etc.” Not one makes sense. Meanwhile, the “evolution” argument is just one big “affirms the consequent” logical fallacy, while Paley’s excellent argument has never been overturned, and an intuitive intelligent design detector can be used to easily disprove “evolution”. Is there a need for any more proofs? Not really. Are there any other proofs? You bet. Take entropy for instance…
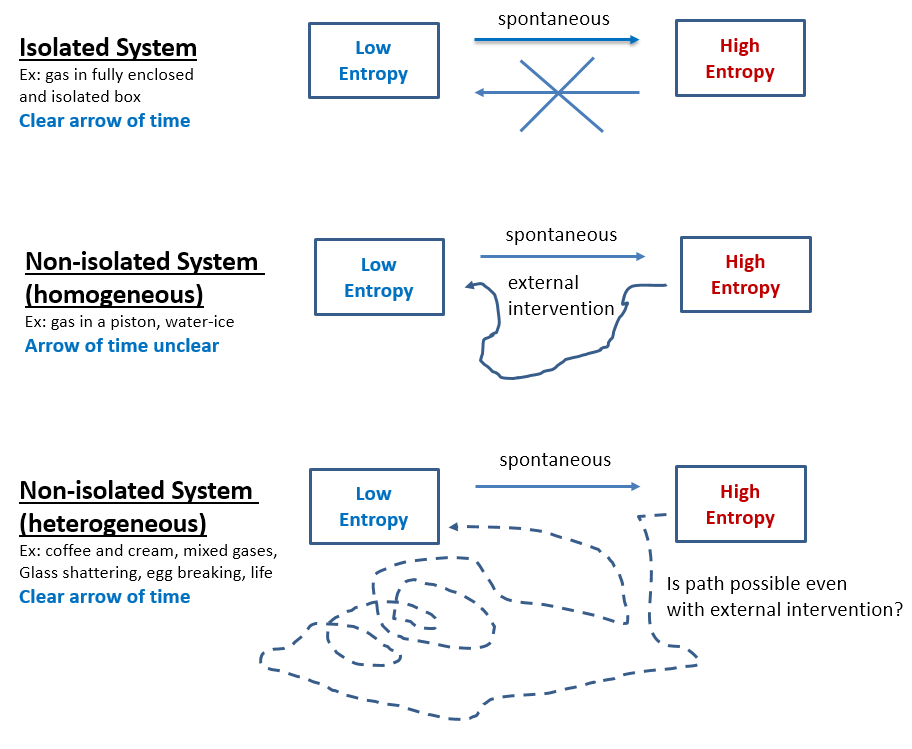
Figure 1
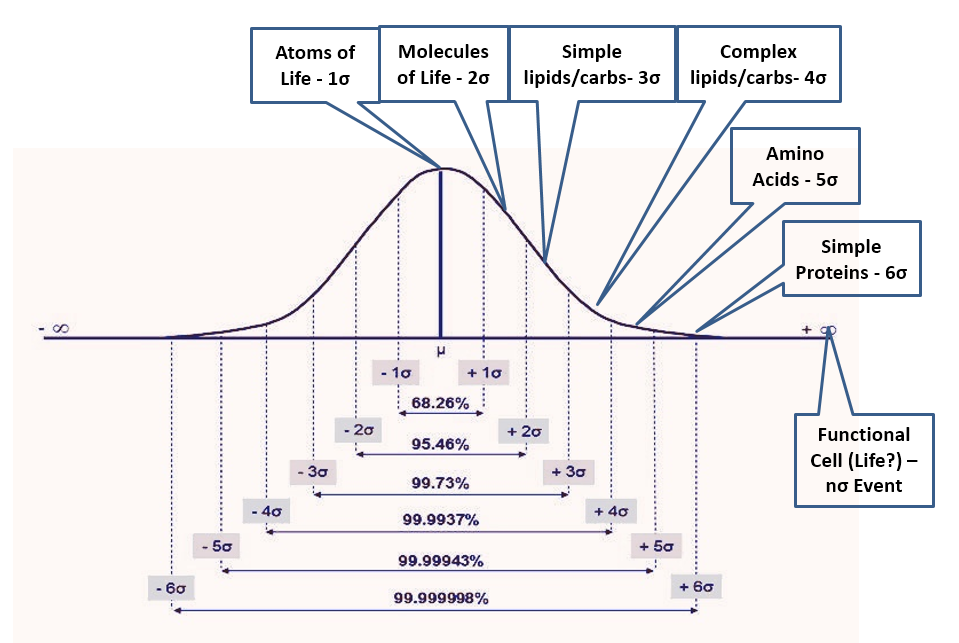
Figure 2
- Second Law of Thermodynamics shows that a spontaneous process cannot also revert spontaneously. This is because spontaneous processes always increase the system’s entropy. A uniform gas in a chamber will accumulate in a corner only with external intervention and spontaneous chemical reactions can only revert if external work energy is applied. Current models of entropy assume the gas particles in a chamber to be independent (sometimes represented as pebbles on a Go board) and explains their never observed convergence on one side of the chamber as only due to that particular microstate having a very low probability(*). However, gas particles always interact with each other (Brownian motion) while pebbles do not. Thus, a reliable way to know that entropy of a system increases is if work energy could be obtained when transitioning from the low to the high entropy state while energy is always required for the reverse process.
- Total entropy of an isolated system can never decrease. Entropy is currently assumed just a statistical law. Thus, if N molecules are in an isolated system (box), the number of microstates associated with j of them being in one half while N-j being in the other half is Ω = N! / (j!*(N-j)!). If N is small, fluctuations seem possible, but before N increases to anything measurable, the probability of fluctuations rapidly decreases to nil. Furthermore, even these theoretical fluctuations, as improbable as they are, might be impossible since the statistical view does not account for molecular interaction observed as Brownian motion and as gas resistance to compression and expansion. Better fundamentals or statistics, either way entropy will never decrease spontaneously in an observable system (Fig 1.a).
- Decreasing entropy is not the reverse process of entropy increasing. That is why a broken egg coming together is easily identified as unreal and a reversed movie of its real shattering. The known laws of physics are the same forward and backward (time-reversal invariance), therefore the reverse shattering process of an egg would not violate any law, but only because these laws are always idealized. Supposedly, if just the right forces are applied to the broken pieces, the egg will come together. In reality this is impossible, and not because the unbroken egg is a highly unlikely microstate, but because entropy increase is not directly reversible even in non-isolated systems. This irreversibility holds for all heterogeneous systems, including life which is perhaps the most heterogeneous system of all. Entropy increase is directly reversible only for homogeneous systems and only if in a defined space. For instance, an expanding gas in an ideal piston creates a force that, when reversed, compresses the gas back into its original state. However, a solid cube of ice can be easily melted by increasing the temperature, but the original ice cube will not reconstitute by lowering the temperature, hence this process too is irreversible despite the cube of ice being homogeneous (Fig 1.b). As far as heterogeneous systems, even separating two mixed gases is way different than the original mixing process, hence mixing is irreversible (Fig 1.c). Entropy decrease is not only different, but also much more complex than entropy increase which is usually spontaneous. Abiogenesis is the entropy-lowering reverse of the biologic decay process, and therefore – if at all feasible – much more complex than adding chemicals and energies.
- Once in equilibrium, a “primordial soup” does not change spontaneously. Life is metastable – it requires certain forms of energy to sustain and spontaneously decays when it no longer receives that energy as well as after the end of the normal lifespan of the organism. It was hypothesized that random fluctuations can spontaneously create compounds and structures given enough time. Abiogenesis, as a reverse-decay process, cannot simply be an outcome of Brownian motion of the chemicals mix because a perpetual motion machine powered by decay and abiogenesis cycles would violate the ‘conservation of energy’ principle. Experimentally, one can confirm that chemical blends in static equilibrium never transition spontaneously into a different equilibrium state (this includes oscillating reactions after the settlement period).
- A “primordial soup” cannot generate life even if energy is applied. It was hypothesized that abiogenesis can be a product of tidal pools, deep sea hydrothermal vents, and the undersurface of ice caps where persistent and abundant energy is available in the form of thermal and electrochemical gradients. Indeed, energy can throw systems off balance and create all kind of chemical compounds and physical structures. However, as the energy applied increases, a complexity limit and hence a dynamic equilibrium is reached where molecule destruction offsets their creation and, if even more energy is applied, molecule destruction dominates, eventually leaving the experimenter with gunk and none of the desired molecules. Miller–Urey and subsequent experiments were not ended because they reached their goal – life – nor because they ran out of energy and materials, but because they reached this dynamic equilibrium, and by adding more of anything would have left them with fewer of the targeted compounds. The amino acids obtained were not the end product but the intermediate between the original molecules and the useless gunk that was the product of the Maillard reaction caused by the energy applied to the system. More complex molecules (and maybe life itself one day) can be created by intelligent designers adding targeted compounds and energies. Then “why can’t natural processes somewhere somehow just mimic the intelligent designer in this vast and almost timeless universe?” The better question is: “why insist on natural processes when the model to be mimicked is that of the intelligent designer?”
- If natural processes were capable of generating life, the environment would be full of intermediate bio-compounds. Life is so complex that laboratories have no hope of replicating it in the foreseeable future. However, if abiogenesis were an outcome of natural processes, the cell structure would be produced only from subsystems and complex biomolecules that in turn would depend on simpler molecules down to H-C-O-N, the atoms of life. A “primordial soup” capable of generating life, thus must contain all intermediate compounds from the atoms of life to the most complex biomolecules and subsystems in an ever-decreasing ratio as complexity increases. Not knowing anything about how this process would work (or even if possible), the most reasonable assumption is a normal distribution of outcomes with life being an n-sigma event (with n unknown) while the availability of the atoms of life being a 1-sigma event and anything else falling in between (Fig 2). Many x-sigma events would be required for each (x+1)-sigma event, with a good first approximation given by the normal density function. Thus, the 2-sigma event could be the basic molecules of life (water, methane, etc.), and we would expect only one of these events for every seven of the 1-sigma events. This approximation would further yield (in one scenario) 1/7 fewer molecules of life than atoms of life, 1/17 fewer simple lipids and carbohydrates molecules (3-sigma) than of molecules of life, 1/43 fewer complex lipids and carbohydrates (4-sigma) than 3-sigma events, 1/110 fewer amino acids (5-sigma) than 4-sigma, 1/291 fewer simple proteins (6-sigma) than 5-sigma, 1/771 fewer complex proteins (7-sigma) than 6-sigma and then – rule of thumb – 1/1600 (8-sigma), 1/3800 (9), 1/9100 (10), 1/22k (11), 1/52k (12), 1/126k (13), etc. fewer of each additional sigma event than previous event where 8+sigma being (this scenario) nucleic acids, short chains, long chains, organelle subsystems, organelles, other critical cell components and finally the fully functional biologic cell – the n-sigma event which is not quite life but good enough for this analysis. Then how can we test this?
- Apart from life itself, the complex molecules of life are nowhere to be found in the universe. To test the ‘natural processes’ hypothesis of abiogenesis, one must observe the intermediate components of life in nature and in the ratios estimated above (or from another reasonable estimate). In addition, one must observe the spontaneous transitions (aided by energy) from simple to complex even if not all transitions are observed at once. Earth is “polluted” with life down to the deepest ocean trenches, therefore the first focus is the extraterrestrial space where, too bad, the largest confirmed interstellar molecules have a maximum of 13 atoms (apart from C60/C70 fullerene). Back on earth we see all intermediate components, but only within life itself. Outside of the cells, aside from the simplest biomolecules, we only see products of decomposition that are never in the ratios associated with abiogenesis, meaning we never see increasing molecule complexity in decreasing ratios resembling anything reasonably expected. Abiogenesis is not happening due to the irreversibility of the entropy increase and for the same reason egg breaking, butter melting, gas mixing, etc. are not reversible processes. Humans can only create a few of the complex molecules, although most always aided by life itself, and even then the power of synthetic biology is severely restricted. The more complex, the harder these molecules are to obtain and the faster they decay instead of spontaneously combining with one another to form even more complex compounds and ultimately life.
- Miller–Urey style abiogenesis experiments are ill conceived, hence doomed from the beginning. To be more specific, they are only good for PR (public relations) given the irrelevant “organic compounds” created that raise the hopes of the believers. Trying to obtain an automobile from scratch by mixing chemicals and energy, qualifies the person attempting as delusional and the one selling such vision as charlatan. So why would those attempting the same with life – which is infinitely more complex than an automobile – not also be labeled charlatans and delusional? Abiogenesis experiments belong to the Reverse Engineering category of processes and, when done right, they are very different than Miller–Urey. Their starting point is never some “primordial soup”, but the most advanced compounds available, preferably already organized in working subsystems. Swapping organelles or parts within organelles, exposing organisms to various environments, attempting to revive dead organisms, substituting engineered subsystems and so on are part of the hard work with long tradition and already being done in medicine and many industries for other purposes than to prove abiogenesis. If and when someone will be able to reverse the decaying and dying processes, we will know that abiogenesis is possible as an act of Intelligent Design creation. To confirm abiogenesis as an “unguided process” we would have to observe reverse-decay and reverse-dying processes happening in nature, not in a lab. Yet 2nd law proves this impossible.
- Is abiogenesis not feasible because it was a unique event? If true, abiogenesis would be a “materialistic miracle” and furthermore not just one, but a long series of “materialistic miracles” since a long series of – so far unknown – events are needed to get from atoms to the simplest organism. Yet one of the tenets of materialism is “no miracles” showing the inconsistency of the materialistic “unique event” assertion. And of course, physics and chemistry transformations are never unique. And even if entropy allowed for abiogenesis, the laws of life do not follow from any priors (physics, chemistry, mathematics). Life has a drive to survive and leave off-springs which entails harm avoidance, immune system, metabolism, food seeking, homeostasis, growth, reproduction, and body structure. Without these, any cell would start decaying the instant it was formed as in fact it does as soon as it no longer is alive. Despite having lasted almost since the formation of The Earth, life is metastable – one knock and it dies and then decays. This is unlike other negative entropy machines that can be restored (rebuilding proportional with the damage).
- Other considerations.
- “Dissipation-driven adaptation of matter” (J. England, MIT) claims that life is inevitable because life “absorbs and dissipates more energy from external sources” leading to faster entropy increase. However, there is no law that entropy has to increase faster. In addition, most of the entropy in the universe is captured by black holes with life having a nil contribution to that entropy.
- Some claim they have obtained “protocells” that seem to mimic real cells at least in part. However, “protocells” are to biological cells as fool’s gold is to real gold.
- “Kolmogorov complexity is lowest at low and high entropy and high in the middle hence life is supposedly inevitable (S. Carroll)”. However, life is not complexity. Life is much more than snowflakes, vortices and chemical reactions (candle burning). And most certainly, life is not the complex swirls of cream mixing into coffee on a journey from low entropy to high entropy (both having low complexity). In addition, unless very specific external action continues to be applied to maintain those patterns, they soon disappear like in sand dunes exposed to shifting winds. The patterns therefore do no “arise”, but are created by an external force.
- “Gradients of energy in deep vents are responsible for abiogenesis”. But all organisms from these exotic places are very similar to any other ones found elsewhere, hence all likely have the same origin. In addition, no free floating organic compounds (aside from decay byproducts) have been found there to suggest ongoing abiogenesis. And, aside from the simplest molecules, no spontaneous transitions from x-sigma to (x+1)-sigma bio complexity has ever been observed around these deep vents either.
- Of course life does not violate 2nd Organisms do conform to 2nd law when they decay as soon as they die. In addition, as observed by Erwin Schrödinger, “the increase in entropy from turning our low-entropy food into our high-entropy waste is greater than the local decrease in entropy from making the well-ordered structures within our bodies”. Nothing special so far – a refrigerator does the same: creates a zone of low-entropy while the entropy of the whole system increases and for as long as it’s fed energy.
- Randomness can theoretically account for any bizarre occurrences including Paley’s watch and F. Hoyle’s 747 in baby steps if enough time is given. But no such event was ever observed. In addition, breaking down the unattainable complex system into a combination of simpler components, each with higher probability of occurrence makes it no easier as the probabilities of all subsystem have to be multiplied to get back to the complex final assembly.
- Some claim that life itself prevents abiogenesis by ingesting all intermediate molecules spontaneously formed, but this can be easily prevented in sterile labs. In addition, all complex intermediate molecules observed outside of cells are due to decomposition, not abiogenesis.
- “Evolution” corollary number 1. If abiogenesis is impossible as an undirected, natural process, then whoever is responsible for abiogenesis is also responsible for the biologic landscape past and present, therefore “evolution” is also impossible as an undirected, natural process.
- “Evolution” corollary number 2. It is easy to verify that nothing ever “evolves” in the nonliving nature. Life is said to be “just chemistry”. These two combine to: nothing “evolves” in the living either. Solar systems, geographical features, fluid eddies, chemistry, snow flakes, etc. all go through their life cycles, and all are different from each other, but the life cycles of the newer entities are no more “evolved” than the life cycles of the ancient ones.
- “Evolution” corollary number 3. Presumably, “evolution” has not ended. And if ongoing, then one must see the normal distribution of the different transitioning organisms (the intermediary), just as we would see if abiogenesis were true. If humans evolved from monkeys and “evolution” is ongoing, then humans must still be in transition especially since the human population is one of the largest of all mammals and, the more individuals, the more “evolving” opportunities. The older Darwinists replied with a hierarchy of races. But that reply is not only fashionably repugnant, but also false and, amazingly, contrary to [at least] the Abrahamic religions that have always known better.
- In conclusion, abiogenesis is nothing more than the decay process running backwards, therefore easily visualized, yet impossible according to the second law of thermodynamics. In other words, “evolution” is nothing more than imagination run wild. Expecting abiogenesis to be within reach if only the proper forces and chemical compounds were added is as wrong as expecting the broken egg to come back together if only the proper sequence of forces were applied to the broken pieces.
Summary:
- A spontaneous process cannot revert spontaneously.
- Mixtures will never ever spontaneously separate per second law.
- Decreasing entropy is not the reverse process of entropy increasing and also much more complex.
- Once in equilibrium, a “primordial soup” does not change spontaneously.
- A “primordial soup” cannot generate life even if energy is applied due to dynamic equilibrium.
- If natural processes were capable of generating life, the environment would be full of intermediate bio-compounds.
- Apart from life itself, the complex molecules of life are nowhere to be found in the universe.
- Abiogenesis experiments belong to the Reverse Engineering category of processes.
- Miller–Urey style abiogenesis experiments are ill conceived, hence doomed from the beginning.
- Abiogenesis unique event conflicts with the “no miracles” clause of materialism.
- Even if entropy allowed abiogenesis, the laws of life do not follow from any priors (physics, chemistry, mathematics).
- “Evolution” corollary number 1 – no abiogenesis, no “evolution”.
- “Evolution” corollary number 2 – no “evolution” in the inert and “life just chemistry”, then no “evolution” in the living.
- “Evolution” corollary number 3 – no intermediate “evolving” entities, no “evolution”.
- Being a decay process running backwards, abiogenesis is as impossible as a broken egg being reconstituted by the “proper sequence of forces”. “Evolution” is also nothing more than imagination run wild.
(*)R. Penrose “The Emperor’s new mind”; PBS SpaceTime “The Misunderstood Nature of Entropy”; Sean Carroll “From Eternity to Here”, etc.
Links:
Abiogenesis: The Faith and the Facts
James Tour: The Mystery of the Origin of Life
Chirality, Maillard – caramelization, characterize the structure at every step:
https://creation.com/why-the-miller-urey-research-argues-against-abiogenesis
https://evolutionnews.org/2014/06/squeezing_the_l/
https://www.ncbi.nlm.nih.gov/pubmed/21422282
Entropy of a box of molecules
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2253472/ https://en.wikipedia.org/wiki/Normal_distribution#Cumulative_distribution_function
http://physics.bu.edu/~redner/211-sp06/class-engines/class25_secondlaw.html
https://www.quora.com/How-quickly-is-the-entropy-of-the-sun-changing
https://www.thoughtco.com/how-many-atoms-in-human-cell-603882
https://www.amazon.com/Mysteries-Modern-Physics-Sean-Carroll/dp/1598038699
https://en.wikipedia.org/wiki/File:Elements_abundance-bars.svg – abundance in the solar system
https://en.wikipedia.org/wiki/Miller%E2%80%93Urey_experiment
https://en.wikipedia.org/wiki/Proteinogenic_amino_acid
https://en.wikipedia.org/wiki/Artificial_gene_synthesis
https://www.scientificamerican.com/article/a-new-physics-theory-of-life/
https://en.wikipedia.org/wiki/Dissipative_system
https://pubs.usgs.gov/gip/dynamic/exploring.html

You may believe this yourself. Dawkins called this “virtuoso believing”.
Therefore, according to you, cars are built by crash-demolition-compression-rusting going backwards like a movie running backwards. Then why were you so insistent that entropy wouldn’t allow it? You have a very weird way of admitting defeat. You rather deny entropy altogether.
So you think you were built from molecules-to-cells-to-you by reverse-decay? After so many times insisting that reverse-decay cannot happen? Well, of course. If cars built themselves in reverse-movie fashion, why not you? All that stuff about people eating, breathing, the ensuing gametogenesis, the encounter between sperm and zygote, etc, must be nonexistent in your fantasy world. You were built by reverse-decay and that’s it.
Anyway, then we’re done. Thanks for admitting defeat. Even if in this extraordinarily weird form. I liked your other admission better though. When you said that energy is not an issue for abiogenesis.
I win. In the weirdest way imaginable, but I win.
Oh dear. This is not going to end well for nonlin.
I will deal with the “n-particle system” stupidity first. Your equation in para 2 refers to the entropy of mixing. You probably don’t realize it, nonlin, but you have assumed that your n-particle system and your 2n-particle system are at the same temperature.
The smaller system may have more entropy, if it is warmer. How much warmer? You are NOT going to like the answer, I fear.
To explore this question, let’s think about your delightful “entropy decrease for one pound of biomass from molecules to cells (as in abiogenesis) “. Tough to know how you are going to quantify that. I vaguely remember a creationist falling into the same beartrap here a couple of years ago: the comparison was with a rat before and after being subjected to a Waring blender. This seems close to your “molecules to cells” change (in reverse), if it’s a really good blender. I think the living rat had more entropy than the homogenized rat, simply because it was slightly warmer.
To reiterate, any ‘organizational’ entropy decrease you might care to postulate wrt abiogenesis will be tiny compared to the entropy decrease of cooling magma. Even after normalizing for weight (which, btw, is totally bogus…)
If you go strictly ‘informational’, and melted enough ice to account for the informational content of the human genome every second, after 31.5 million seconds (a year) you would have melted 1.5 micrograms of ice.
There’s a reason they are called the Laws of Thermodynamics: it’s about heat. And, to return to your original claim, abiogenesis and it’s decrease in entropy is dwarfed by daily local decreases in entropy like ponds freezing.
Does anything?
😀
If your claims were scientific it would be possible to publish them Nonlin. Given that you go against everything scientists have understood so far, you’d find resistance, but, if, as you claim, your shit is scientific, and irrefutable, it would take only a few rounds of discussion with reviewers to get it in. After all they’d be renowned scientists. Scientists who can see a good scientific insight when it’s well presented to them. Hell, if you were right, nature would fight to get your articles published there first. The news and views section would read something like:
“Kindergartener finds astounding evidence that evolution never happened” or “Kindergartener finds that entropy, against everything we thought we knew, is an unsurmountable barrier to abiogenesis.”
After all, conferences all over the world. Everybody trying to meet the kindergartener who put Einstein to shame. Everybody wanting to meet you first. To shake your hand. Their only desire to see if your brilliance is contagious.
You’re wasting your time here. Run to nature! If you’re from the US run to science magazine. I’m sure they’ll be happy to beat the English journal in finding you. That you’re a kindergartener won’t make a difference. You obviously have us fooled here that you’re an unbeatable intellect, seasoned by years and years of experience and study. So, they won’t even notice until it’s published. You’ll be able to say “take that adult retards!”
😀
So a river under constant flow is in ‘dynamic equilibrium’? So what? You can drive a turbine from it. It is not in thermodynamic equilibrium.
What, in order to accept as a reasonable explanation the notion that primitive life would be uncompetitive against modern life, we’d have to create it? What a heap of shite.
Untrue. The ‘driver’ for a meteorite falling to earth, for example, is gravity. That is spontaneous, in thermodynamic terms. The ‘driver’ for a redox reaction is (partly) the differential shielding of charge in atoms with more than 1 proton and 1 electron, leading to an asymmetry in the electrostatic force, which tends to rebalance at a lower-energy state, if accessible. Many such reactions are thermodynamically spontaneous, either in isolation or when coupled.
You are saying here that inorganic redox reactions do not happen, unless I show you an example. Your ignorance is astounding.
Many different atoms and molecules in many different physical arrangements.
No. That’s the sort of thing someone who thinks abiogenesis is ‘decay running backwards’ might say.
No, it isn’t. How many ‘pounds of abiogenesis’ are we talking, here? You can seed gallons of medium with a single bacterium. That’s how much ‘entropy decrease’ you need to get the process going: that contained in one cell. Subsequent organisation is taken care of by growth and replication. ie if the ‘end of abiogenesis’ (and the ‘beginning of life’) is a replicating entity, the amount of ‘entropy decrease’ you need to kick it off is that amount or less. A drop in the ocean.
You show no such thing. Therefore nothing.
If the topic is ‘entropy forbids abiogenesis’, a proper understanding of the energetics of entropy change would seem to be a basic requirement. And you are nowhere near such an understanding.
Not really. They come and go, in a similar manner, for similar reasons.
So ‘dynamic equilibrium’ is necessary to sustain things? Haha. You’ve shot yourself in the foot. The ‘equilibrium’ state is necessary to sustain life, but is (somehow) inimical to the origination of life. Yuk yuk.
Nonlin.org,
Given your noted difficulties with the concept ‘100%’, this isn’t all that solid a self-endorsement.
Nonlin.org,
Haha, the vaguest definition is also the ‘most scientific’. Good one. Where did this come from? It sounds a bit amateurish, for someone claiming to speak ‘the language of science’.
As I illustrated, the term covers a wide variety of processes, only one of which (the chemical one) has the apparent quality of thermodynamic equilibrium. The rest require work to be done to maintain. And, being non-equilibrium in the thermodynamic sense, simultaneously have the capacity to do work – some of the work put in, you can get back. So I don’t see any of these other applications of that vague definition helping your crusade.
They cannot be. Besides wrong being too kind a word for most of your claims, half of your claims contradict the other half. Contradictory claims cannot both be true.
You understand nothing.
WRONG. Search “number of microstates formula”.
WRONG AGAIN. There’s no temperature in the entropy formula:
S=k_{b}\ln\Omega
S = entropy
k_{b} = Boltzmann constant
\ln = natural logarithm
\Omega = number of microscopic configurations
No need to. We can compare.
FALSE. Ice is homogeneous, hence any water molecule can be anywhere in the system whereas cells are heterogeneous and molecules MUST BE in a limited number of places. Fewer microstates, lower entropy. Pound for pound.
I showed my proof. Where’s yours?
Prove “total bogus”!
Thee is no “information” in physics. Not in the elementary particles, nor anywhere else. Information is abstract. Shannon’s entropy is a metaphor. And BTW, Shannon’s “information” is actually “data”. He was solving ‘data transmission’, not whatever auntie had to say. If you knew anything about “theory of information” that is.
Don’t you see how illogical your claim is? If that were the case, how come no one can produce abiogenesis when pond freezing is so mundane?
No. To have ice you need a considerably ordered structure of water molecules. Individual water molecules need to be close together and have a specific orientation to form hydrogen bonds.
Then I’m right about dynamic equilibrium. As always! Stop mumbling and say so!
What “primitive life”? We’re talking about the building blocks of life. If vent holes produced proteins, DNA, RNA, lipids, etc, we should see them being produced and then eaten by “modern life”. And in a sterile lab there is no “modern life” to even eat those compounds so abiogenesis should be a breeze. If those vent holes do NOT produce those compounds, then obviously NO abiogenesis there. WTF is your problem that you don’t understand something as basic as this?
False again. Gravity is always there if the meteorite falls or not. The DRIVER is whatever propelled the meteorite in the collision orbit.
Whatever reaction is spontaneous in one direction, needs a driver, hence is NOT spontaneous in the other direction. Fact. Decay – spontaneous, abiogenesis – NOT spontaneous.
You understand nothing. Perhaps willfully.
So your task is to build a cell. What are your raw materials again if not the same ones a cell is composed of? Be specific.
Retard question. Pounds of raw materials that will be transformed in the SAME number of pounds of cells.
Yes. Make ONE cell! 10 nanograms… since you don’t understand “pound” as figure of speech.
Abso-fucking-lutely! As dumb as they were, even Miller-Urey got this basic fact in their foolish Frankenstein attempt. Why don’t you? Equilibrium is inimical to change: https://en.wikipedia.org/wiki/Chemical_equilibrium . Furthermore, life itself is designed to maintain the equilibrium. We call that homeostasis. And when you have homeostasis, you don’t need to adapt so much. So no “evolution” (not that adaptation is in any way same as “evolution”). It all fits together perfectly!
Other complete nonsense was ignored. On the plus side, you got two likes. Because science is a consensus thing, you see?
Allan Miller,
Take it up with those that define it so, and with those that accept that definition. I like it and it fits 100% the case. This is not the credit card company, so “your opinion (call) is NOT important to us”.
Yes, yes. But they are fully interchangeable. The molecules in the cell are NOT.
Me? It’s you who implied that cars are built by a “movie-running-backwards” process, and went on to ask for experimental evidence that you were not built from a reverse-decay process, as if you thought you were. If that was not your intention, then you should learn to read for comprehension before attempting to give answers. That would also help you with all of those mistaken notions you have of almost everything you “present,” trying to pass it for knowledge.
Don’t you see how illogical your claim is Nonlin? That entropy is not a problem doesn’t mean that we know everything about the conditions that can lead to abiogenesis. It just means that entropy is not the problem.
I’d suggest that, besides reading for comprehension, you took a good course in logic. Reading for comprehension first, otherwise you’ll waste your time with the lessons about logic.
‘Right’? By far the commonest use of the term is in reversible chemical reactions, as I stated. You said that was FALSE. Now you’re declaring yourself ‘right’ because there are other usages, completely unrelated to entropy and your thesis; usages in which there is no thermodynamic equilibrium.
Here’s the bottom line: use of the term ‘dynamic equilibrium’ does not mean that no work can done by such a system. So, regardless whether you see yourself as ‘right’ or ‘wrong’, you’re wrong.
The result of abiogenesis. Keep up.
Ask my wife.
Aha! So it’s not ‘clear and specific’. Nonetheless, gravity has a large part to play. If a projectile were aimed straight at the earth, the entropy at the moment of impact would still contain a component due to gravitational attraction, added to that due to its initial relative velocity.
You’re just declaring it so. You don’t actually know if there is an abiogenetic environment with negative ΔG or not. It certainly isn’t ‘decay backwards’.
I understand that inorganic redox reactions take place. I’ve taken (and passed!) exams in this stuff and everything! Where did you go, Dunning-Kruger U?
I don’t know. But it is not certain that the requirements for the process to commence are solely those present in modern cells. Still, your thesis is that ‘entropy forbids it’, which is not a statement about specific materials.
You just need one ‘cell’, or primitive replicator. The rest of the ‘pounds of material’ come from replication and growth, which you agree do not trouble the laws of thermodynamics.
Sure, colour me confused. I can’t tell when you are ‘speaking the language of science’ and when you are using ‘figures of speech’.
Oh, good grief! Now you are parading my definition! The one you declared FALSE! Vents aren’t in chemical equilibrium. There is no forward/backward reaction to which the balance applies. Additionally, even chemical equilibrium gains a direction if there is consumption of products or refresh of reactants – ie, if the system is open. Have fun trying to argue that a vent spewing into an ocean is a ‘closed system’.
Ugh, this is hopelessly confused. All possible applications of the term ‘equilibrium’ cobbled into one grotesque mess.
Whyever not? Indeed why isn’t homeostasis itself an adaptation?
And who are they?
That a vague definition covers a multitude of specific situations does not make those situations interchangeably identical.
It’s an interesting challenge to read between the lines of mangled prose and the petulant schoolyard taunts to try and understand the true nature of the misunderstanding, which is often hidden.
Here I believe we are seeing a fun version of thermodynamics that is not, in fact, new. This Creationist Law of Thermodynamics is the fairly popular Configurational Law of Thermodynamics (CLoT). Under this CLoT, energy flows are irrelevant. All that matters is some form of ‘configurational complexity’. Increases in CC are forbidden by this 2CLoT; strangely, these CLOTs are unable to explain why heat cannot of itself pass from one body to a hotter body.
Now, this Law would unfortunately prohibit replication and growth, so nonlin has come up with the added stipulation that it is complexity per pound that cannot increase.
Because he only thinks of macroscopic configuration, and not energy flows, he cannot see the difference between steady state and equilibrium, and references to rivers and turbines go straight over his head.
Hence his use of the binomial equation as his “entropy formula” and the delightful claim that
Depends on how we define “so much.” Homeostasis takes a lot of adaptations, from tiny to large, in order to be kept. It’s not an unresponsive state, otherwise it would be easily disrupted. Your body must respond, meaning adapt, to changes in the environment in order to keep its balance. Given the constancy of responses, I’d say that if you have homeostasis is because you have been adapting a lot.
You keep using “equilibrium” in different situations to mean that practically nothing is happening, even when you accompany the word with “dynamic” as if the word “dynamic” didn’t mean anything at all. I know you’re desperate, you’ve been contradicting yourself time and again, and it’s been too easy for us to point and laugh, which you truly hate. But this is plainly ridiculous Nonlin. Truly, learn to read and check your ego at the door. It makes you do the most openly dishonest “moves,” and that doesn’t help your “cause,” even if your cause is just to uplift your ego, rather than defend your supposed faith. We won’t feel “taught” by you any time soon if all yo’ve got is equivocations accompanied by tantrums. Sorry. It doesn’t work that way.
DNA_Jock,
Yes, this seems to be the case. When one points out the absence of consideration of energetics, one is directed to Point 5, where energy belatedly rears its head. But then continues to be ignored in all subsequent discussion, which seems to be all about molecular configuration, and non-interactive reversibility. Shannonesque, IOW. Again, I blame ‘order’! 😎
Allan Miller,
I do enjoy all this talk of energy being applied in order to overcome/circumvent/deal with the 2LoT and allow the creation of complex order.
It is the fact that energy is removed that allows all those local decreases in entropy.
The heat sink is absolutely required. You only need an energy source if you want the bloody thing to keep going.
I think that’s the wrong focus. The 2nd law is not circumvented, it runs its course. Things happen because of the 2nd law. The 2nd law is the “guarantor” that things will happen. The answer to the question: “How do you reconcile entropy and evolution?” is not “it’s an open system” or anything like that, because the question is loaded. It assumes that entropy is a problem. The answer is “there’s nothing to reconcile, entropy runs the show, entropy is the reason there’s evolution.”
Entropy,
True. Both my growth and my eventual decay are entropically ‘forward’ processes. I eat pies; eventually bacteria will eat me. There is no appeal to temporary exceptions to the 2nd Law – I’m not that bothered about statistical mechanics, and am happy to sign up to an effectively deterministic version with no uncompensated reversal.
Even the expulsion of waste is not the bottom of the entropic well – we have extracted some of the energy, but there’s bags left. Whole ecosystems gorge on it, gaining local entropy decrease in their turn while ‘decaying’ the material. With that in mind, if abiogenesis was reverse decay, eating would be ‘reverse crapping’! My intake and outflow, after all, are arguably in a state of ‘dynamic equilibrium’, according to the Nonlin Doctrine.
Horses for courses.
I do see your point, but would describe things differently. If I were asked to explain the awesome power of anabolic chemistry, I might go with “entropy makes things happen”. But the supposed problem is almost always couched in terms of [local] decreases in entropy being a contravention of 2LoT. In which case, all you need is a heat sink.
I think a more general and accessible way of putting it is to say that “energy flows downhill”, a la Flanders and Swann. And it’s pretty obvious that if energy did NOT flow downhill, then there would be no chemistry and, as I like to put it, we would not be having this conversation.
Entropy is a book-keeping convention (albeit an unreasonably successful one) that brings together all the different ways that energy flows downhill. Imbuing it with agency, as in “the Entropic force” or “entropy makes it happen”, is an invitation to misunderstanding, or to getting trolled. By the same token, describing the Law of Large Numbers as a ‘force’ that drives any sample’s average towards the population average leads to similar confusion. Amongst the same people, even. 😉
DNA_Jock,
to-may-to, to-mah-to
Entropy,
I’m a Brit living in the USA. These things matter.
Been watching The Crown this week.
Allan Miller,
You’re in denial and are now going in circles. Don’t know if anything is worth addressing for the 100th time. Perhaps a few:
If “certain”, produce your proof!
Because homeostasis is as primordial as DNA, proteins, metabolism, etc.
Allan Miller,
Like I said. Denial.
I’m not sure what you’re dreaming there.
Anyway, here’s an interesting presentation that marries energy quanta with the microstates formula: https://ocw.mit.edu/resources/res-tll-004-stem-concept-videos-fall-2013/videos/governing-rules/entropy/
The only mistake I see is that some of those microstates are not compatible with the macrostate. As in all energy concentrated in a corner of the system.
WTF? Then do explain why Miller-Urey applied energy in their foolish attempt. And if energy were removed, do explain how come the cell has higher energy than the raw materials?
Tell you what: since you’re such fan of cooling magma and melting ice, how about I race your “cooling magma”/”melting ice ” powered engine while I use good old fashioned fossilized BIO-fuel? Haha.
Sure is according to the time reversal invariance. You might say you’re full of it… were it not for energy and gas flows.
DNA_Jock,
Cheap and very bad philosophy. Haha.
Meanwhile, abiogenesis is nowhere in sight. Don’t you ever wonder why? Because it’s not possible, that’s why. Because entropy forbids it.
To not speak of all the claims from last time you abandoned. Perhaps you recognized how weak they were.
Nonlin.org,
I’m still wondering what the next step is. My suspicion is there isn’t one. The Ancient Mariner will continue to harangue us here ‘cos nowhere else will put up with him.
Nonlin.org,
You have descended into a level of incoherence well beyond anything you’ve written before. Amazing.
P.S. I win!
Entropy,
I do feel kinda bad; but the inability to get the point can be interesting —
Nonlin.org:
Nope. That’s not the mistake. The mistake is that you have not noticed that the number of energy quanta in the system matters. In a rather impressive manner. For instance, in your video, Prof Lienhard notes:
How many different ways can the quanta be distributed if there is only three in a bar? Compare that result, squared, with 4 x 56. This is why “heat cannot of itself…”
DNA_Jock,
Tell me about it.
What do you think was the energy that dissipated as those more complex compounds were formed in the experiment?
Astounding the amount of ignorance concentrated in this single question. I don’t even know where to start.
P.S. I win!!!
How the fuck would you know what I have not noticed? How do you know my statement is not more profound than your primitive understanding? Go ahead now that you have been warned. Put more thought into this. A lot more! Let’s see if you’re capable.
DNA_Jock,
And where’s your ice powered engine? When do we go racing?
Amazing! One guy disputes well-established concepts. Another challenges Boltzmann. All think entropy decrease is as trivial as entropy increase and that life SELF-sustaining is the same as life creation. Some guys think A-to-B is somehow not the reverse of B-to-A (then what is?). Another guy thinks you decrease the entropy by removing energy from the system (perhaps thinking life is just hot gas). I keep hearing from dead men and other third parties as if those have anything to say about this study. And on and on and on… The nonsense never stops.
Nonlin.org,
The next step?
That would violate Entropy’s Law.
What does that have to do with anything? I’d really like to know. Furthermore, I’d like to know what it has to do with microstates, macrostates, statistical mechanics or thermodynamics?
Mung,
It’s how many ways microstates sum to the same macrostate.
Even if I can’t, why does it matter? I don’t even know what it means for energy to “spread out,” much less why it matters.
Can you illustrate a real-world system in which energy isn’t already “spread out”?
These are nonsense questions meant to confuse people like Nonlin. 🙂
Nonlin.org,
How do I know you are not profound, nonlin? Because, nonlin, you wrote this:
and this
and a hundred other amazingly wrong things.
Here’s the entertaining thing, nonlin: I have given you numerous opportunities to prove me (and my low opinion of you) wrong. All you would need to do would be show the mathematical ability of a reasonably smart High School student. (Most recently with this “How many different ways can the quanta be distributed if there is only three in a [four atom] bar?”)
Strangely, you repeatedly “choose” not to humiliate me with your competence, and instead opt for grade-school taunts. The conclusion is inescapable.
I’m not sure what you mean. Are you saying that the number of ways that the energy quanta can be distributed is the sole determinant of the number of microstates in a macrostate?
By the way, I think this topic falls under statistical mechanics. If that’s not what you’re talking about let me know. 🙂
Just think of all the ways they can be configured!!!
https://en.wikipedia.org/wiki/Configuration_entropy
Mung,
My law is that I always win.
Indeed.
I wouldn’t take this one as amazing. You do this all the time, it was to be expected you’d do that here as well.
You didn’t really challenge Boltzmann, you’re just too illiterate to think of checking the formula beyond the names of the components of the equations.
You think that? because I don’t. I’m just convinced, by everything that surrounds and happens around me, that local entropy decrease is energetically trivial, more importantly if we’re talking about the kind of decrease primitive life would consist of, compared to current life.
That must be you, because I don’t even think that life is a “creation.”
Well, if you want to believe that you were made by a reverse-decay process, despite you have insisted that’s not possible, be my guest. You lose either way. As for me, I understand that the paths cannot but be different given the second law. The keyword, for the millionth time, is “path.”
(I keep wondering about that figure and the bent and twisted lines, what do they represent if not paths that differ from the one going towards entropy increase? Why would one of them be labeled “path” if it’s not a path? If it’s a path, why would the line be all twisted if it’s just the same path going in reverse?)
Energy dispersal drives local decreases of entropy,.
Yours certainly is.
Ha! “study”! Hahahahahahahaaaaaaaa! Holy crap! So funny! Anyway, you must believe that Boltzmann is still alive.
One thing we can agree about. But don’t worry Nonlin, your nonsense is very entertaining.
I have a thermodynamic system consisting of two dice. Each die has six faces. Each face of each die is painted with one or more spots The number of spots on each die is between one and six with no face on a given die having the same number of spots as any other face on that same die.
How many “macrostates” are there? How many “microstates”?
Are you saying I should stop ignoring Nonlin? Is the entertainment value really worth it?
So what. You always win.
Mung,
How warm are these dice?
It’s being a lot of fun for me. Once I understood that Nonlin is uneducable, I started to appreciate the entertainment value of her/his nonsense.
But that doesn’t mean it would be equally fun for you.
Why does that matter? Does how warm they are somehow depend on the energy quanta per number of dots? Somehow the die is warmer on the face that has only one dot than it is on the face that has six dots?
You’re making my point. Thanks.
Oh dear, Mung.
In case you forgot, you asked.
That’s gonna depend on the temperature. I was really expecting you to play your hand better than that. Clearly, you did not spend your time away honing your act.
Right back at ya. My point being, the configurational entropy is minor, compared with the distribution-of-energy-quanta entropy. Check out nonlin’s MIT professor.
When you asked:
You should have read the following two sentences. Here’s the whole paragraph.
Let me know if you don’t understand the Flanders and Swann song.